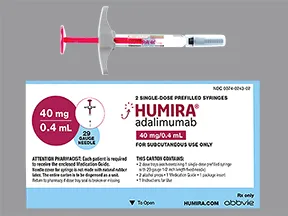
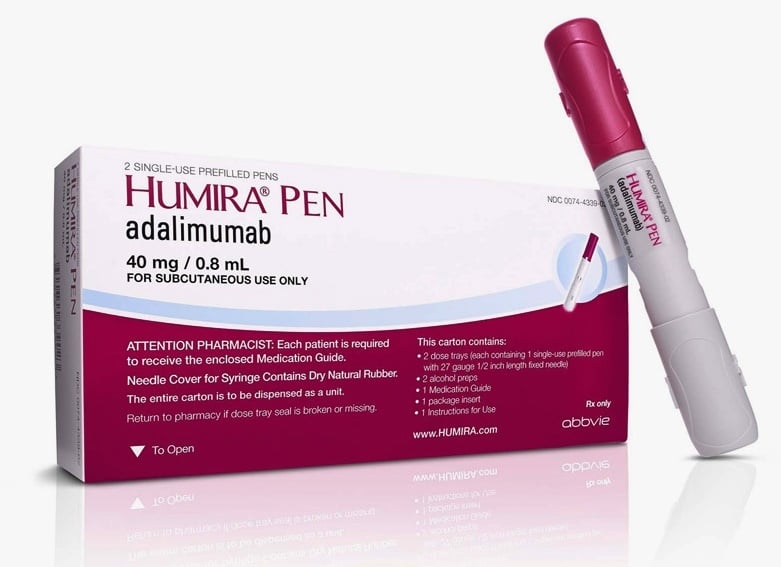
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE

Humira (Adalimumab Injection Solution for Subcutaneous Administration): Uses, Dosage, Side Effects, Interactions, Warning
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE
Similar Pharmacokinetics of the Adalimumab (Humira®) Biosimilar BI 695501 Whether Administered via Subcutaneous Autoinjector or Prefilled Syringe (VOLTAIRE®-AI and VOLTAIRE®-TAI): Phase 1, Randomized, Open-Label, Parallel-Group Trials | Rheumatology ...

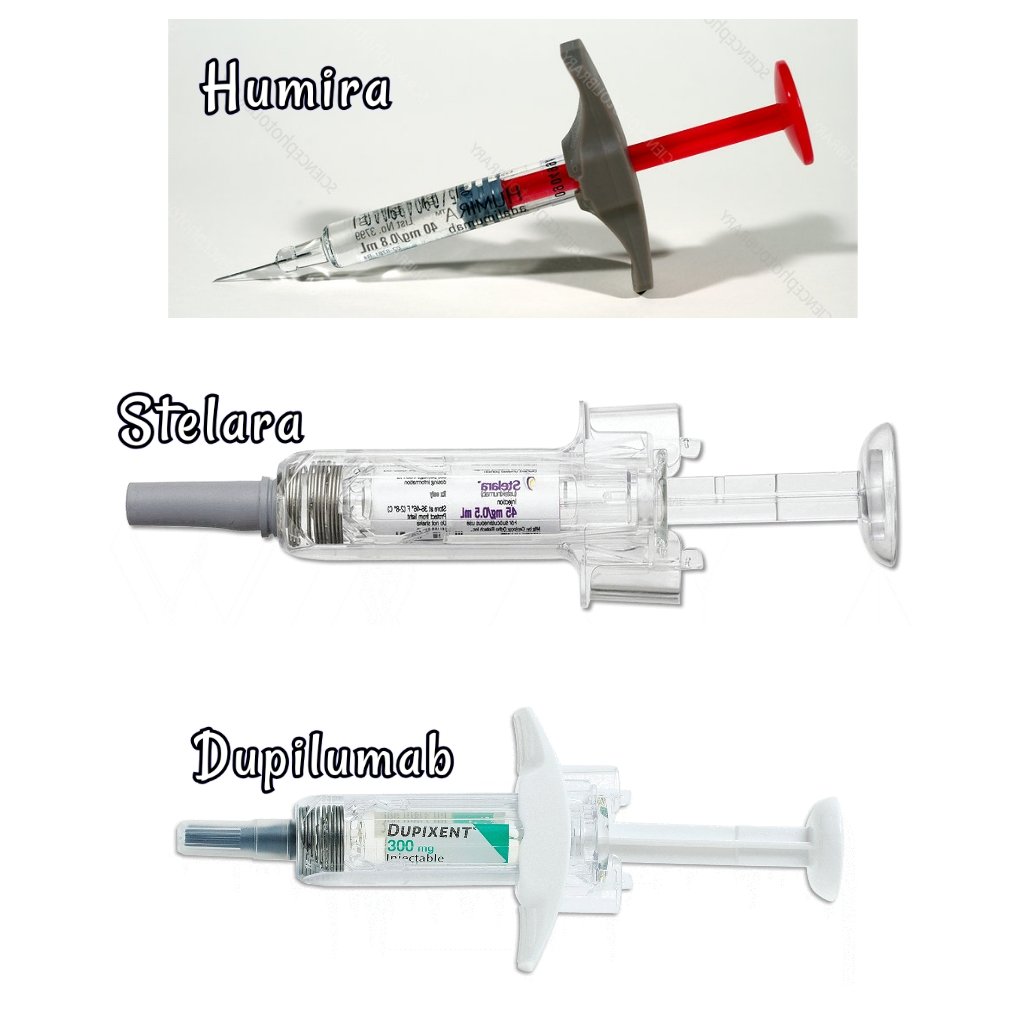
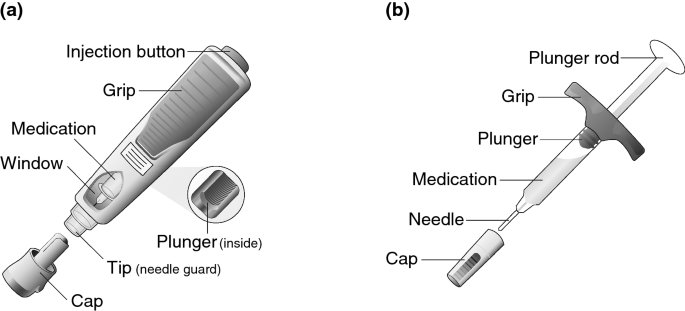
Components of the adalimumab (HUMIRA ® ) Pen before, during and after... | Download Scientific Diagram