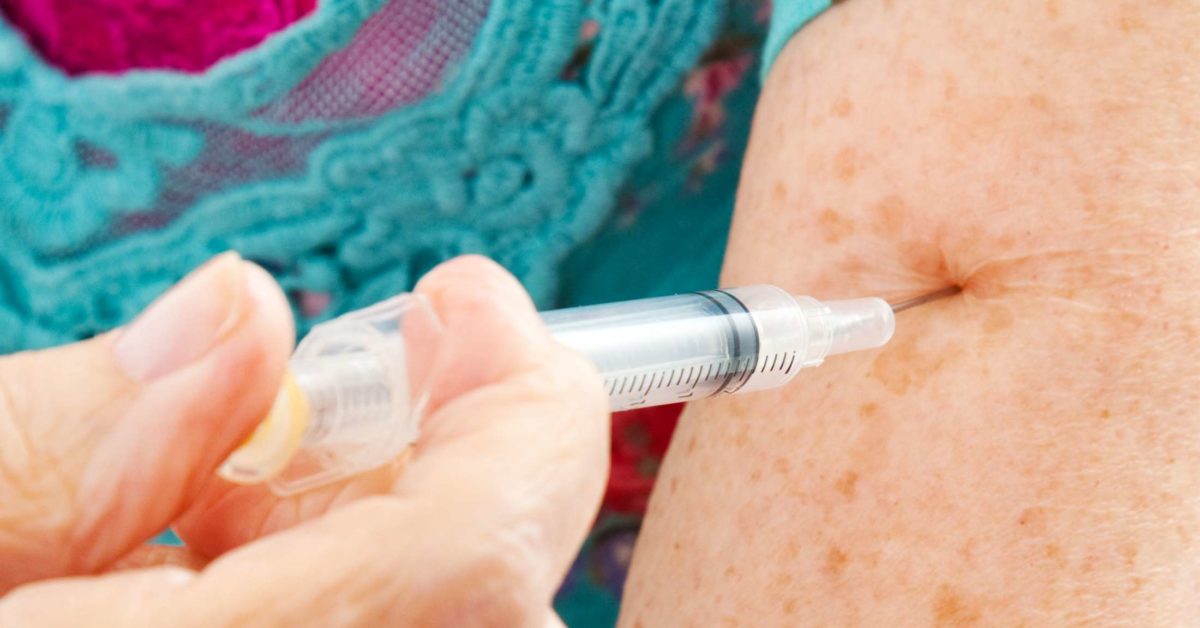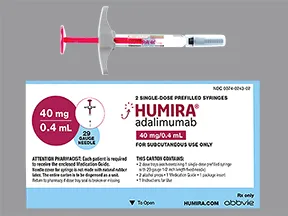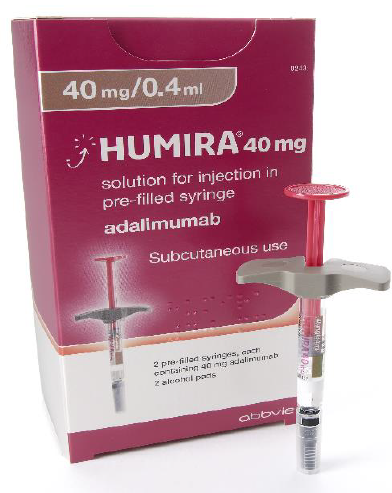
Choosing between available adalimumab products – SPS - Specialist Pharmacy Service – The first stop for professional medicines advice
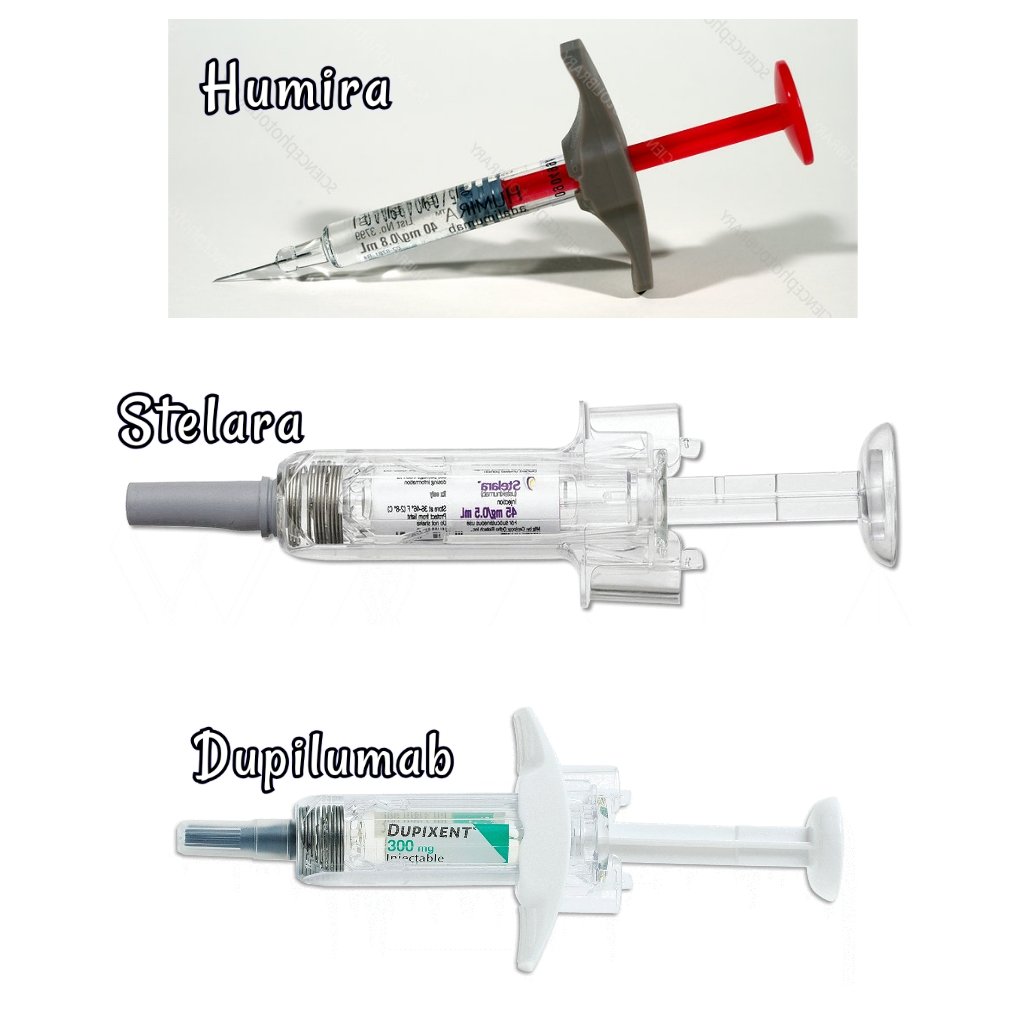
Natalia on X: "1/2 Anyone else with #crohns #eczema struggling with safety syringes? I injected #humira for 10yr, great control but since changing to #stelara & #dupilumab have to wrestle with big,
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE
INSTRUCTIONS FOR USE HUMIRA® (Hu-MARE-ah) (adalimumab) 40 MG/0.8 ML, 20 MG/0.4 ML AND 10 MG/0.2 ML SINGLE-USE PREFILLED SYRINGE